Reduced Pain
Over the years PEMF Therapy has proven to be effective on both acute and chronic pain. The body is comprised of approximately 70 trillion cells which communicate via Electromagnetic Frequencies. When the body experiences Electromagnetic pulses, it generates micro currents which target the cell membrane modifying and restoring normal function to damaged cells.
Improved Sleep
PEMF Therapy has developed an almost universal reputation for improvement in sleep. People with electrical hypersensitivity often suffer from issues such as: problems falling asleep, early awakening, multiple awakening or long waking hours during the night. PEMF therapy devices have proven to be the most effective in stimulating brainwave patterns to shift from disturbing patterns to patterns seen in normal sleep behavior.
Increased Circulation
The primary function of our magnetic fields in the body is to naturally increase circulation by opening and dilating the arteries and capillaries. When PEMF Therapy is used, a magnetic pulse passes through the cell which enables the membrane to become charged. The cells then separate from each other which helps to promote proper circulation and an increase in cellular oxygen.
Reduced Inflammation
Inflammation is caused by the body attempting to heal itself after experiencing injury. PEMF Therapy helps to reduce the inflammation by charging the cells with a gentle electromagnetic pulse. Once charged, the cells then increase their oxygen carrying ability which promotes healing, reduces swelling and pain. The electromagnetic pulse that flows through the body stops the release of inflammatory mediators and increases blood flow.
 What are harmful EMFs?
What are harmful EMFs? How can PEMF therapy help?
How can PEMF therapy help?
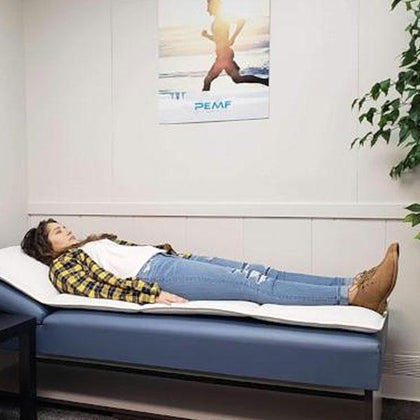












 What are harmful EMFs?
What are harmful EMFs? How can PEMF therapy help?
How can PEMF therapy help?